|
2/29/2024 0 Comments Modern periodic table with details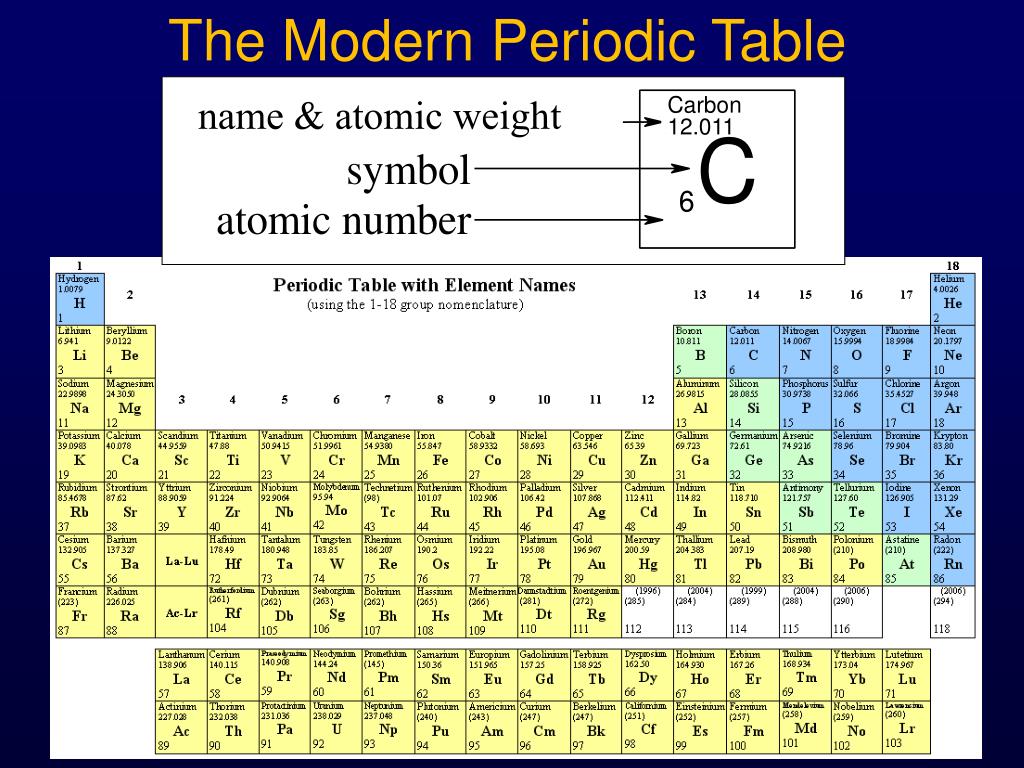 
There are 18 groups and 7 periods in the modern periodic table.Įlements having same number of valence electrons are placed in the same group.The vertical columns are known as groups and horizontal columns are known as periods, in the modern periodic table.Elements are arranged in order of their increasing atomic numbers.Elements in each class share certain fundamental features. There are three different types of elements including metals, metalloids, and nonmetals. Elements in the same group have similar characteristics. Groups are the columns of the periodic table. If we move from left to right across a period, we observe that each element has one more proton than the element before it. The number above each symbol represents its atomic number.Ītomic numbers in the table grow from left to right and from top to bottom. Each element is represented by its chemical symbol in the current periodic table. 
Elements are organized in the table by increasing atomic number. The modern periodic table is used to organize all known elements. Periodic trends are based on the periodic law, which asserts that if the chemical elements are listed in increasing atomic number order (from left to right), then their physical and chemical properties will be a periodic function of their atomic numbers. They result from variations in the atomic structure of elements with their appropriate periodic table periods (horizontal rows) and groups (vertical columns). The electrical configuration of elements determines periodic trends. Periodic table trends are predictable patterns in the properties of periodic table elements. Periodic Trends in the Modern Periodic Table Notice how the atomic numbers in the table rise from left to right and from top to bottom. The number above each symbol in the table represents the symbol’s unique atomic number. It is an abbreviation for cuprum, the Latin word for copper. The modern periodic table looks like the following:Ĭhemistry Formulas Exploring Crystallography Understanding the Solid State Examining the TableĮach element is denoted by a chemical symbol consisting of one or two characters.The first letter of a sign is always capitalised whereas the second letter, if any, is always lowercase.Ĭu, for example, is the symbol for copper. He received his degree in chemistry in 1865, following various illnesses and the loss of his mother. He would study chemistry at his father’s alma mater, the Main Pedagogical Institute (not St. Dmitri’s father died unexpectedly when he was thirteen. Dmitri’s father lost his sight shortly after his birth. He was the youngest of fourteen children still alive. Father of modern periodic tableĭmitri Mendeleev, the father of modern periodic table, was born in Siberia. A new periodic table based on modern periodic laws was constructed by altering Mendeleev’s periodic table. Scientists created various artificial elements. People became more interested in the chemistry of these elements due to their similarity. Scientists now have a comprehensive understanding of quantum numbers and the electrical structure of elements in the periodic table after discovering the fundamental unit of elements.Ĭhemists discovered a resemblance between the 94 naturally occurring chemical elements after learning the periodic law. The number of electrons or protons in a neutral atom is represented by the atomic number. “Elements’ physical and chemical properties are periodic functions of their atomic numbers.” The current Periodic law can be stated as follows: There are 18 vertical columns and seven horizontal rows in total. The modern periodic table is the periodic table in its current form. 
The table is an arrangement of elements arranged in increasing atomic number order. The modern periodic table, often known as the long version, is based on modern periodic law.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
